Netherlands CDMO Manufacturing Consulting
Leverage YCP's global expertise in the Netherlands' CDMO manufacturing sector. By offering extensive services through proven strategies in the industry, YCP guides businesses to thrive in the Netherlands' competitive market. Benefit from our decades-long experience and hands-on support.

YCP in Numbers

With the Dutch pharma sector's export value reaching
€29.3 billion*
in 2024, Netherlands remains a strategic hub for CDMO manufacturing opportunities.
Source: CBS, 2024
YCP CDMO Manufacturing Experts in Netherlands
The Netherlands is rapidly becoming a key player in the biotechnology industry with several technological advancements. This surge is reflected in the booming CDMO manufacturing sector, adapting swiftly to global demands and consistently innovating to meet future industry needs.
YCP, equipped with a team of global professionals and a profound understanding of the biotechnology landscape, delivers unparalleled CDMO manufacturing consulting services. We effectively help clients navigate market entry, overcome growth barriers, and facilitate expansion in the Netherlands' dynamic biotechnology sector, strategically leveraging our deep industry expertise.
YCP stands as a reliable consulting partner for businesses aiming to excel in the Netherlands' CDMO manufacturing market. Our unwavering commitment to addressing complex industry challenges endorses our credibility as a leader in biotechnology consulting. With YCP, clients gain access to strategic insights that foster sustainable growth and success.
Unlocking Potential: Your Path to Excellence with
Netherlands's CDMO Manufacturing Professionals
CRO Lab Optimization
Our innovative strategies in Netherlands' CDMO manufacturing elevate pharmaceutical production efficiency and accelerate time-to-market for groundbreaking therapies.
Bench Productivity
In Netherlands, YCP excels in CDMO Manufacturing consulting, boosting bench productivity through tailored strategies for industry advancement.
Analytical Debottlenecking
Expert CDMO Manufacturing Consulting in Netherlands ensures optimally efficient processes using analytical debottlenecking solutions for operational success.
Our Services
Digital Transformation
Leverage cutting-edge strategies to thrive in digital transformation in the Netherlands.
- Digital Roadmap & DX Strategy
- DX Maturity Assessment
- CIO/CDO Advisory & IT Organization Design
- System Architecture & IT Portfolio Management
- AI, RPA & Intelligent Process Automation
- Big Data & Analytics
- Data Governance & Digital Adoption
- Offshore Development Centre Set-up
Market Research
Uncover industry trends and insights for strategic positions in Dutch CDMO manufacturing sectors.
- Market Landscape
- Market Size
- Industry Structure and Trend
- Competitive Benchmark
- Business Partner Search & Screening
- B2B/B2C Customer Survey
- Consumer Research
Marketing & Sales
Navigate complex CDMO challenges with tailored strategies enhancing your manufacturing in the Netherlands.
- Branding & Brand Management
- Product & Portfolio Management
- Pricing
- Digital Marketing
- E-commerce
- Customer Experience
- Sales Channel Strategy
- Digital Sales
- Lead Generation and Nurturing
- Content Marketing
M&A, Transactions, and PMI
Nurturing revitalized business structures and seamless transitions across the Dutch M&A landscape.
- M&A Strategy & Deal Sourcing
- Buy-Side & Sell-Side Advisory
- Cross-border M&A Advisory
- Commercial Due Diligence & Valuation
- Divestiture & Carve-out Strategy
- Post-Merger Integration (PMI) & Value Creation
- Joint Venture
- Corporate Venture Capital & IPO
Operational Transformation
Leveraging vast industry expertise, YCP optimizes CDMO operations for efficiency in the Netherlands.
- Operating Model Redesign & Organization Design
- Operational Excellence & Continuous Improvement
- Cost Reduction & Business Process Improvement
- S&OP, IBP & Demand Planning
- Asset Performance & Predictive Maintenance
- Sales & Channel Excellence
- Transformation Office & Enterprise Performance Management
Operations
Navigating complex CDMO manufacturing processes in the Netherlands with tailored operational insights.
- Project Management Office (PMO)
- Growth Strategy Implementation
- Operation Transformation
- Manufacturing
- Organization Design
Public Services
Leverage tailored consulting solutions to optimize CDMO manufacturing processes in the Netherlands.
- Economic Development
- Public Policy Development
- E-Governance
Strategy
Strategically leveraging CDMO expertise in the Netherlands, YCP optimizes manufacturing pathways.
- Corporate & Business Strategy
- Growth Strategy & Market Entry
- Business Plan & Mid-Term Management Plan
- Competitive Landscape & Value Chain Analysis
- New Business Development & Feasibility Study
- Strategic Partnership & Channel Strategy
- Organization Strategy & Change Management
- Enterprise & Asset Risk Management
- Strategy Execution Support & Business Turnaround
- Digital Strategy
Supply Chain Management
Seamlessly navigate the complexities of Netherlands' CDMO sector, boosting efficiency and innovation.
- Supply Chain
- Digital Supply Chain
- Procurement
- Logistics
Sustainability
YCP identifies inefficiencies, enhancing sustainable practices within Netherlands' CDMO Manufacturing.
- Sustainability Strategy & Implementation
- Double Materiality
- Decarbonization & Net-Zero Strategy
- Climate Change Risks and Opportunities
- Sustainability Due Diligence
- Supply Chain Assessment
- ESG Reporting
- Investor Relations & Fundraising
- Organization & Governance Design
Navigating CDMO Manufacturing Sector in Netherlands
Limited R&D Resources For Dutch CDMO Companies
- Dutch CDMOs face a significant R&D challenge with only 15% business budget commitment to research. This limitation restricts innovations in drug manufacturing.
- With increasing demands for innovative medicines, CDMOs should look towards forming strategic partnerships or utilizing EU innovation grants to close innovation gaps efficiently.
Regulatory Compliance Could Impede Growth Trajectories
- Navigating detailed EU regulatory frameworks poses a hurdle for Dutch CDMOs, extending project timelines by an average of 20%. These complexities can slow market entry.
- A competent regulatory strategy leveraging digital compliance tools could streamline processes, ensuring smoother approval pathways while controlling costs.
Emerging Technologies Touted In CDMO Advancement
- Technological advancements like AI and automation are growing, capturing 40% of sector investments. These empower more precise and efficient processes.
- Shifting operations to integrate these technologies can reduce error rates in manufacturing by over 50%, enhancing both product outputs and market competitivenessó.
Increase in EU Biopharma Collaborative Ventures
- Biopharma alliances have surged by 30% in the EU for year total collaboration, pointing to beneficial partnerships with Dutch CDMOs focused on niche specialties.
- Enhancing cross-border collaborations adds value to CDMO operations, positioning them strongly in niche areas and increasing foreign investments and trust.
Our Clients
We have worked with more than 2000 companies throughout various industries.












What Our Clients Say About Us
"YCP impressed me most with their ability to get to the truth on some very specific questions, bringing a level of concreteness to the content that I’ve never seen from another consulting firm. Their work provided valuable analysis for decision-making. The team at YCP is efficient, insightful and fearless!"

General Manager for APAC & Director of Global Logistics and Material Handling Business Unit
Intralox
"We are very happy with the projects handled by YCP for Saint-Gobain. The YCP teams have been fully engaged and have shown real professionalism. They understood our needs and completely fulfilled our expectations. Thanks to a step by step process, many interactions were possible between YCP and our company, and their team reacted with the right flexibility and pace. The studies conducted by YCP delivered very practical recommendations and valuable conclusions for us. We definitely recommend YCP. "

Global Marketing Director
Saint Gobain
Results Through Expertise: Case Studies
Learn how we help clients build and implement strategies
that drive sustainable growth in today’s complex business landscape.
E-Commerce Website Development
Client
A Japanese medical equipment manufacturer
Area
Southeast Asia
Expertise Scope
Strategic Marketing
Project Summary
Our client needed support tapping into the Southeast Asian market and building an e-commerce channel. We created a market entry strategy in three countries, developed localised landing pages, and managed client campaigns to gain market access.
Acquisition Support for Medical Device Distributors in Southeast Asia
Client
A well-established European healthcare multinational company
Area
Southeast Asia
Expertise Scope
M&A Target Identification and Due Diligence, Market Entry Strategy
Project Summary
We assisted a European healthcare multinational in acquiring medical device distributors or OTC manufacturers across Southeast Asia. Over 12 months, we shortlisted top targets using strict criteria, initiated direct outreach, and facilitated NDA signings and meetings. A promising prospect in Singapore was identified, leading to a non-binding offer in due diligence. The client then expanded the project to the entire region.
YCP in Media











Our Group CEO Yuki Ishida and Managing Partner Karambir Anand shared insights at the ASEAN–Japan Young Business Leaders’ Summit.

At the Asian Power Summit 2025, our Director Harika G. led a session on Asia’s evolving energy mix.

Singapore Business Review featured Karambir Anand on Singapore’s expanding solar capacity and energy challenges.
Publications

AI in Healthcare: Keeping the Patient at the Center of Innovation
AI is reshaping healthcare by improving efficiency, diagnostics, and personalized care amid rising costs and workforce shortages. Applications span prediction, telemedicine, diagnostics, and robotics across providers, insurers, and pharma. Long-term impact depends on responsible adoption, data quality, and strong governance.

India’s CRDMO Market: Building Momentum through Operational Excellence
India’s CRDMO market is growing rapidly, driven by China+1 strategies, funding access, and policy support. However, scale, cost pressure, and regulatory demands pose challenges. To compete globally, companies will depend on operational excellence, technology adoption, and building scalable, end-to-end capabilities.
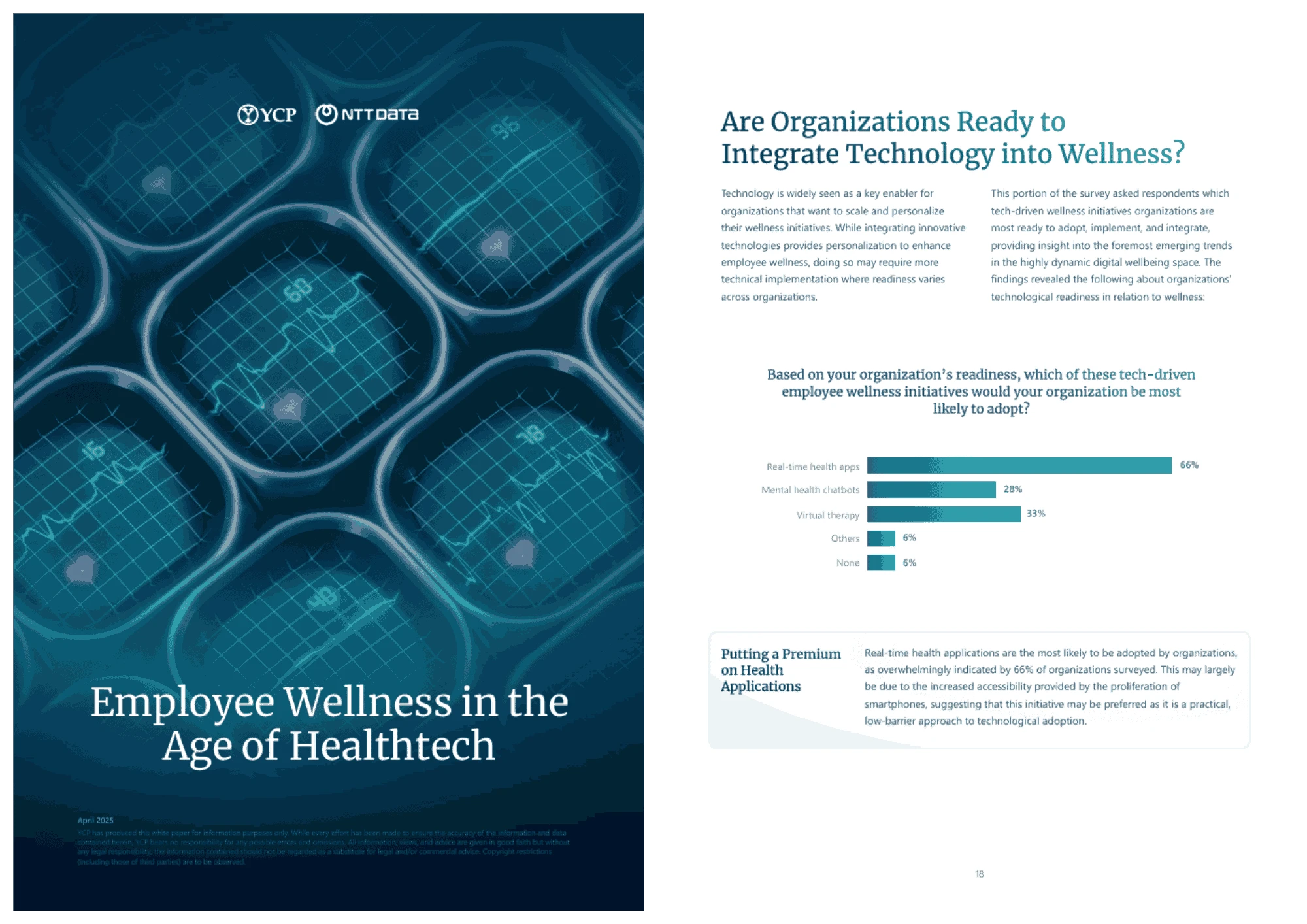
Employee Wellness in the Age of Healthtech
Employee wellness is becoming a strategic priority as workforce expectations evolve. Health technology is shifting programs toward personalized, proactive solutions, particularly across Asia. While data, privacy, and ROI challenges remain, organizations that integrate technology with human-centered design can build more resilient workforces.

Unveiling the Potential of the Halal Industry: Emerging Trends in Southeast Asia
Global demand for halal products is rising, driven by Muslim population growth and broader interest in ethical offerings. Southeast Asia plays a central role, with Malaysia, Indonesia, and Singapore leading in certification, consumption, and trade. Future growth depends on harmonized standards, technology adoption, and sustainability focus.
Meet Our Experts
Establish industry dominance with our experts and partners

Yuki Ishida
Managing Partner
Nationality: Japanese
Region Coverage:
Asia, North America
Past Experience:
Goldman Sachs

Pilar Dieter
Managing Partner
Nationality: American
Region Coverage:
Asia, North America
Past Experience:
Alaris Consulting

Laurent Fihey
Partner
Nationality: French
Region Coverage:
Europe, Asia, Middle East, Africa, North America, South America
Past Experience:
Arthur Andersen, Advancy

Leon Cheng
Partner
Nationality: Chinese
Region Coverage:
Asia, North America, South America
Past Experience:
EAC, DHL

Abhisek Mukherjee
Managing Partner
Nationality: Indian
Region Coverage:
Asia, Europe, North America
Past Experience:
SDG, Accenture Strategy

Paul Archer
Partner
Nationality: British
Region Coverage:
Indonesia, Singapore, Hong Kong, Malaysia, Thailand, , Philippines, China, India, Australia, United Kingdom, United States, United Arab Emirates
Past Experience:
Cummins Inc

Darren Kenneally
Partner
Nationality: British
Region Coverage:
Southeast Asia, USA, United Kingdom, Brazil and India
Past Experience:
Dartington Crystal

Mridul Karkara
Partner
Nationality: Indian
Region Coverage:
India, United States, Europe, Southeast Asia
Past Experience:
THB, Deloitte
YCP Group is the leading professional firm in Asia empowering excellence through diverse expertise:
- YCP
- YCP Auctus
- YCP Consus
Talk to Our CDMO Manufacturing Experts
Arrange a session with our experts to explore how YCP can assist you in addressing your business challenges.
YCP Group is the leading professional firm in Asia empowering excellence through diverse expertise:
- YCP
- YCP Auctus
- YCP Consus


